This topic takes on average 55 minutes to read.
There are a number of interactive features in this resource:
 Biology
Biology
 Science (applied)
Science (applied)
People have used the enzymes in yeast for thousands of years to make bread and alcoholic drinks. But since the 1950s, enzyme technology has advanced in leaps and bounds. We now know how to use microorganisms to produce enzymes that we can isolate and use.
Increasingly enzymes are used in isolation from the microorganisms that produce them. Extracellular enzymes are commonly used – they are easier to isolate and use than intracellular enzymes. As most microorganisms only secrete a small number of extracellular enzymes they are relatively easy to isolate and use – and they are usually tougher than intracellular enzymes, being less sensitive to specific pH ranges and temperatures.
However, because there is a much greater range of intracellular enzymes they are increasingly being used in biotechnological processes in spite of the disadvantages. For example glucose oxidase in diagnostic tests, asparaginase in cancer treatment and penicillin acylase for antibiotic conversions.
There are a number of advantages to using isolated enzymes instead of whole microorganisms:


Isolated enzymes from calves’ stomachs have long been used for a long time in the production of cheese, but now the use of isolated enzymes in industry has become much more widespread and varied, including in the production of wine.
Producing isolated enzymes is expensive. When free enzymes are used they are often lost at the end of the process. Increasingly isolated enzymes are also immobilised – the enzymes are held stationary in an inert support system whilst they catalyse the reaction. There are many advantages to this process:
Disadvantages of using immobilised enzymes include the cost of the immobilising process and that sometimes the enzymes are less accessible once they have been encapsulated. There are a number of different ways of immobilising enzymes:
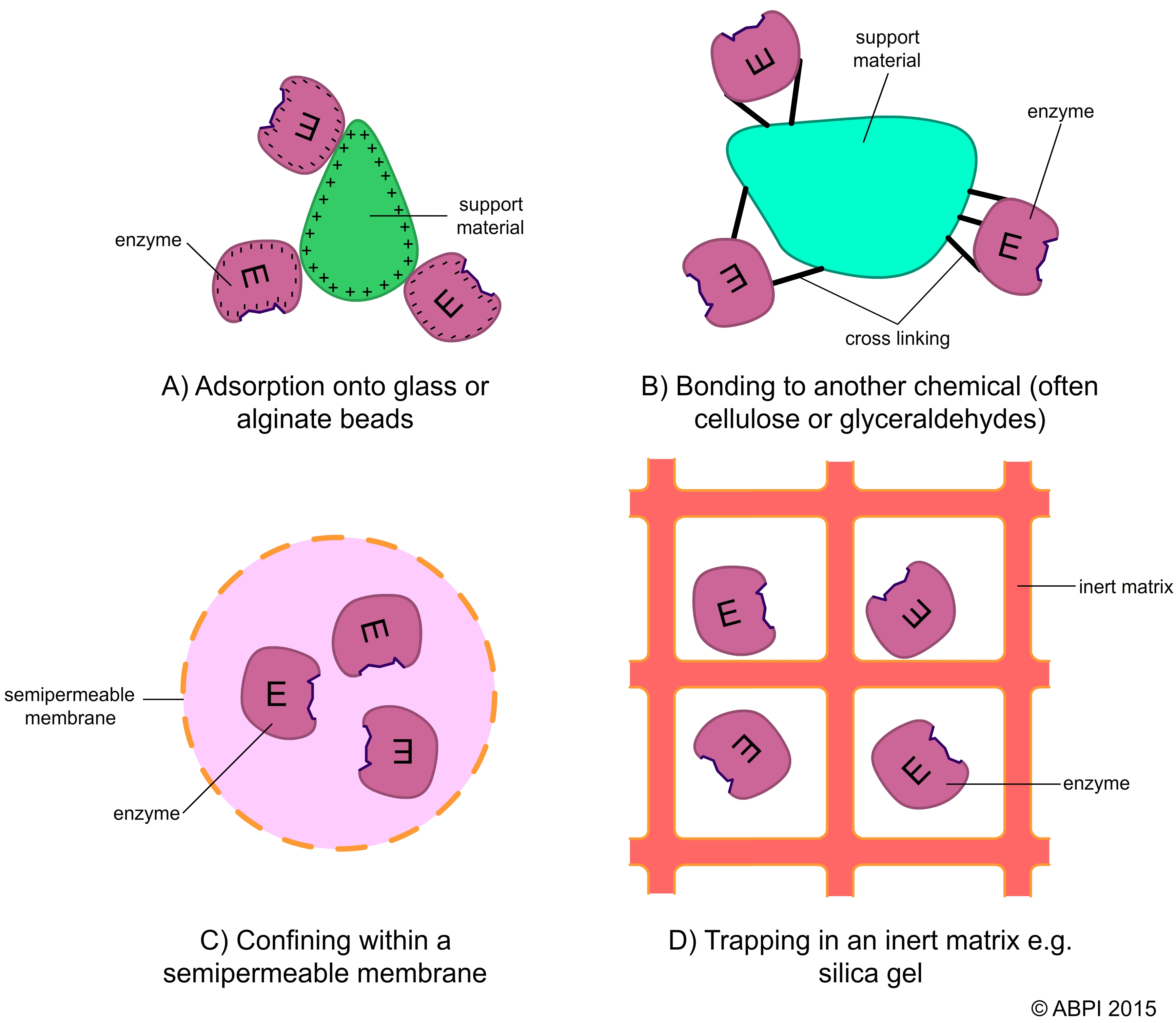
Methods of immobilising enzymes – each has advantages and disadvantages
In the 1960s, the first biological washing powders appeared. It was a revolution – using proteases in detergents meant that protein-based dirt in clothes was broken down by the enzymes, and washes could be carried out at lower temperatures, reducing the use of electricity. Once people realised that boiling clothes was no longer necessary - in fact it made the detergents work less well as it denatured the enzymes - biological detergents became very popular.
Unfortunately the number of people suffering allergic reactions to the detergents also soared. This included both customers and people working in the factories producing the detergents. After a lot of negative media coverage, enzymes were removed from detergents.
Research showed it was the enzyme powder touching the skin or being inhaled by workers that caused the problems. The enzymes were immobilised by being encapsulated. No adverse effects on either the environment or the workers were observed after the enzymes were encapsulated. They were reintroduced and soon biological detergents became very popular again. Now the capsules include lipases and amylases as well as proteases and they are a very large section of the domestic detergent market.
|
Encapsulated enzymes in a biological detergent |
Sales of protease enzymes before |
